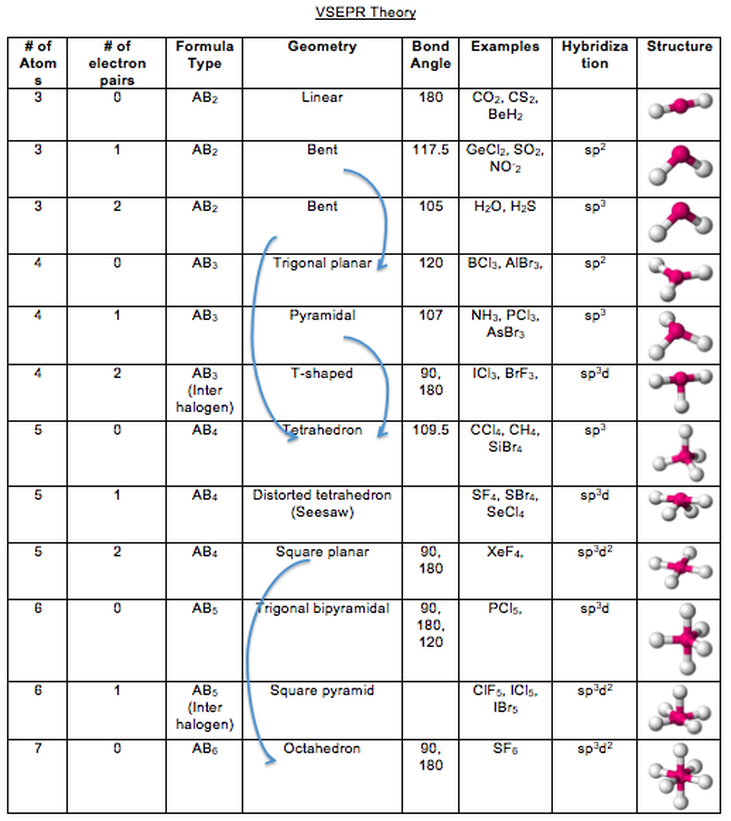
The linear molecular geometry of HCN is important in understanding its chemical properties and reactivity. As a result, the molecular geometry of HCN is linear, with the carbon atom in the center and the nitrogen atom and hydrogen atom on opposite sides of the molecule. However, the molecule is not flat, as the lone pair of electrons on the nitrogen atom exerts a repulsive force on the bonding electrons, causing them to push away from each other. The VSEPR theory predicts that these regions will arrange themselves in a trigonal planar geometry around the carbon atom. 
These regions include the triple bond between the carbon and nitrogen atoms and the lone pair of electrons on the nitrogen atom. The HCN Lewis structure shows that there are three regions of electron density around the central carbon atom. The VSEPR (Valence Shell Electron Pair Repulsion) theory is used to predict the shape of the molecule. The molecular geometry of HCN is determined by the arrangement of its atoms and electron pairs around the central carbon atom. Determination of the shape of HCN molecule Examples of nonpolar molecules include carbon dioxide and methane. In contrast, nonpolar molecules have an equal sharing of electrons between the atoms, resulting in no separation of charge and no dipole moment. The polar nature of HCN makes it a useful solvent for polar molecules and also affects its physical properties, such as its boiling point and solubility. The dipole moment of HCN is 2.98 D, which indicates that the molecule is polar. 
This unequal distribution of charge creates a dipole moment in the molecule, where the negative and positive charges are separated by a distance. As a result, the electrons in the H-CN bond are pulled closer to the nitrogen atom, creating a partial negative charge on the nitrogen atom and a partial positive charge on the hydrogen atom. The nitrogen atom has a higher electronegativity value than the carbon and hydrogen atoms, which means that it attracts electrons more strongly. The HCN molecule is polar because it has a separation of charges between the atoms due to the difference in electronegativity values of the atoms involved. Explanation of the polar nature of HCN molecule Double-check the Lewis structure to make sure that all atoms have the correct number of electrons, all formal charges are small, and the total number of valence electrons is correct.Ĭ.A formal charge of zero is ideal, but small formal charges are acceptable. Determine the formal charges of each atom by subtracting the number of valence electrons in the neutral atom from the number of electrons assigned to it in the Lewis structure.If necessary, move electron pairs to form double or triple bonds to satisfy the octet rule. Make sure that each atom has an octet of electrons, except for hydrogen, which can have two. Check the total number of valence electrons.In HCN, place the remaining six electrons around the nitrogen atom. 
Start by placing electrons around the outer atoms, then the central atom. It states that each atom should have eight electrons in its outermost shell. Distribute the remaining electrons around the atoms to satisfy the octet rule.In HCN, connect the carbon atom to the nitrogen atom with a triple bond and to the hydrogen atom with a single bond. Connect the atoms in the molecule using single bonds to form a skeleton structure.Identify the central atom in the molecule, which is the atom that forms the most bonds.For HCN, hydrogen has one valence electron, carbon has four valence electrons, and nitrogen has five valence electrons, making a total of ten valence electrons. Determine the total number of valence electrons by adding the valence electrons of each atom in the molecule.To draw the HCN Lewis structure, follow these steps: The hydrogen atom is bonded to the carbon atom through a single bond, with each atom sharing one electron. The HCN Lewis structure consists of a triple bond between the carbon and nitrogen atoms, with each atom sharing one electron to form the bond. In this structure, the hydrogen atom is bonded to the carbon atom, which is in turn bonded to the nitrogen atom. The HCN Lewis structure is a representation of its molecular structure that shows the arrangement of its atoms and the sharing of electrons between them. The HCN Lewis structure is important in understanding its chemical properties and how it interacts with other molecules. Hydrogen Cyanide is a colorless, highly poisonous gas that has a faint, bitter almond odor. It consists of one hydrogen atom, one carbon atom, and one nitrogen atom. The chemical formula for Hydrogen Cyanide is HCN.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
